EXPERT SERVICES
Q-Metrx, Inc. has extensive experience in assisting with all phases of clinical trials for drug development and proof-of-concept studies in both healthy volunteers and sensitive patient populations. Q-Metrx has its own recording systems and sensors, as well as a complete qEEG analysis, interpretation, and reporting service.
Our staff — including experienced registered EEG and PSG technologists, sleep specialists, Board Certified neurologists, and PhD trained experts in ERP/EEG analysis and neuroscience — offers a complete solution for your CNS needs. We are experts in neurophysiological methods specifically designed to assess the safety, tolerability and effects of new compounds on CNS activity.
EEG
- EEGs: Screening, Safety and Long-term EEGs available with hyperventilation and photic stimulation .
- Quantitative EEGs (qEEGs)
- Event-Related Potentials (ERPs): P50, P300, Mismatched Negativity (MMN), Pre-Pulse Inhibition (PPI), Faces, Filters and more
Sleep
- Polysomnography (PSG)
- Multiple Sleep Latency Test (MSLT)
- Maintenance of Wakefulness Test (MWT)
- Actiwatch
More

- Pupillometry
- Eye Tracking
- Experimental Design
- Development and execution of Statistical Analysis Plans (SAPs)
- Summary Reports
Electroencephalography (EEG)

Electroencephalography (EEG) is a non-invasive method of recording real-time, spontaneous electrical activity of the brain and is the cornerstone of Q-Metrx services. EEG is a valuable tool for research and clinical trials, as it offers high temporal resolution (on the order of milliseconds), is relatively mobile, and is tolerant of subject movement, which is not true of other brain imaging techniques.
Pharmaco-EEG: Essential Information in Early Drug Development
With the aid of the pharmaco-EEG, essential information can be obtained in early drug development regarding CNS penetration and efficacy of new substances as follows:
-
- ✓
Evidence for substance penetration (bioavailability) in the CNS on the basis of PD changes. In addition to the classic PK parameters (e.g., tmax, Cmax, AUC and t½), EEG/ERP parameters can be used to demonstrate time- and dose-effect correlations.
- ✓
-
- ✓
Proof of CNS effects of a substance by comparison of quantitative EEG (qEEG/ERP) changes between pre- and post-dose measurements.
- ✓
-
- ✓
Characterization of psychotropic properties of a substance by comparison with psychotropic class effects of marketed drugs. Classic psychotropic drugs show typical pharmaco-EEG profiles, thus allowing assignment of new substances in many cases.
- ✓
-
- ✓
Determination of new substance’s therapeutic window. The dose at which a new substance can be statistically distinguished from placebo, and at which it displays a psychotropic effect in comparison with a reference preparation, is often a therapeutically effective dose.
- ✓
-
- ✓
Proof of adverse CNS effects of non-psychotropic substances (e.g., sedation produced by anti-hypertensive drugs or anti-histamines, changed convulsion threshold due to antibiotics).
- ✓
- ✓
Determination of the bioequivalence of two different galenic dosage forms by quantitative comparison of CNS changes.
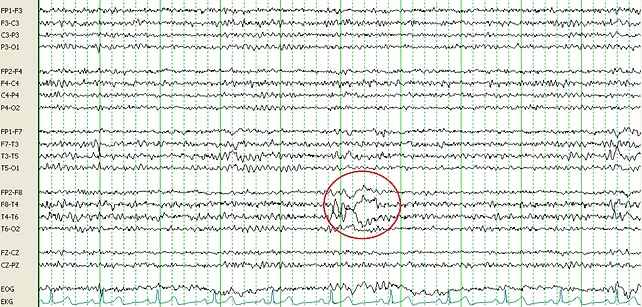
Safety EEG
Safety EEGs are clinical EEGs that are read by a board-certified neurologist and can be used during
a clinical trial to help assess safety and tolerability of a novel compound.
Safety EEGs can be used as an admission tool to exclude subjects with underlying seizure potential,
or they can be used during a trial to ensure that no EEG abnormalities are found after dosing.
qEEG
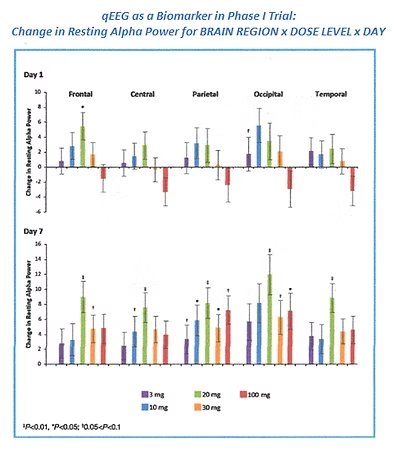
Quantitative EEG, or qEEG, involves digital EEG recording under standardized conditions, removal of
extracerebral artifact, and extraction of specific properties of the signal (e.g., amplitude or
coherence). Results can be shown graphically or numerically where subsequent statistical processing
can be performed.
Quantitative analysis techniques can be used for identification of biomarkers, documentation of
penetration into the nervous system, pre vs. post drug comparisons, exposure-response analysis, time
trend analyses, source localization, and evaluation of patterns of connectivity.
ERP
Event-Related Potentials, or ERPs, are elicited brain responses (measured via EEG) to a specific
sensory stimulus. Common modalities involve auditory or visual stimuli to produce responses that
are averaged over many trials which produce various components of interest.
ERP paradigms can show pre-cognitive, automated processes as well as explicit cognitive processing
even in the absence of behavioral change or subject attention to the stimulus. ERP component
abnormalities in clinical research have been shown in conditions such as: Alzheimer’s (and other
dementia types), Schizophrenia, Parkinson’s Disease, Multiple Sclerosis, and other neurological
disorders.
ERP Paradigms use in past clinical trials include:
- Contralateral Delay Activity
- Implicit Reward-Biasing Task
- 3-stimulus auditory oddball task
- Sensory Gating Paradigm
- Auditory Steady-State Response
- Visual Evoked Potentials
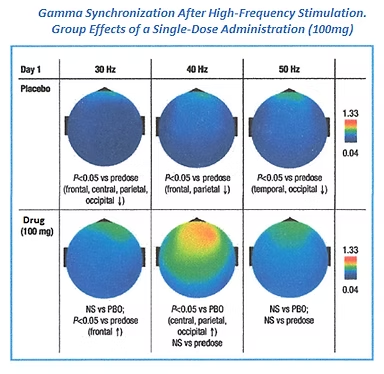
APPLICATIONS
The Q-Metrx Staff includes highly experienced sleep technicians with extensive experience in developing custom protocols for sleep studies with various applications, including:
- Insomnia: Initiation and Maintenance of Sleep
- Excessive Daytime Sleepiness and Sedation
- Narcolepsy and cataplexy
- Obstructive sleep apnea
- Sleep Devices
- CPAP
- Bilevel
- Dental
- Miscellaneous other devices (e.g., Winx & Provent Therapies)
- Parasomnias: REM Behavior Disorder (RBD)

TECHNIQUES
- Polysomnography (PSG): Overnight attended sleep study
- Multiple Sleep Latency Testing (MSLT)
- Subject attempts to take four to five naps every 2 hours throughout the day
- Allows quantification of drowsiness throughout the day
- Maintenance of Wakefulness Testing (MWT)
- Four to five “quiet periods” throughout the day
- Subject must remain awake in a darkened room
- Actigraphy for monitoring:
- daytime activity
- napping
- lights out/lights on associated activity
Contact Form
